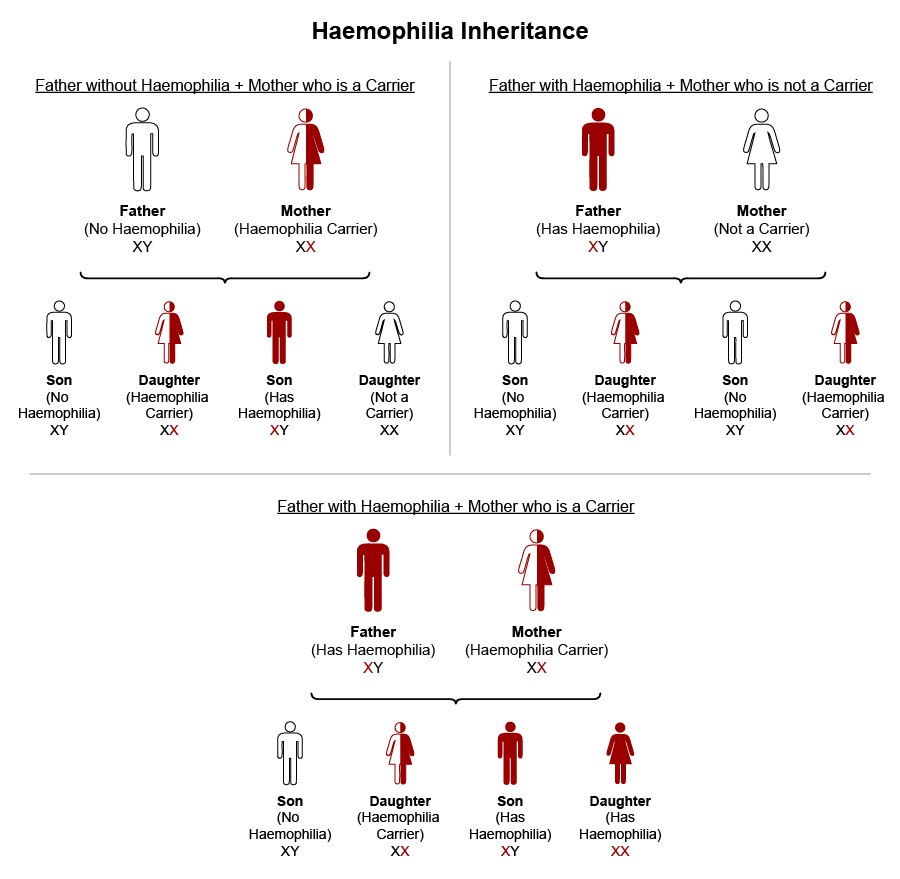
Gene therapy is currently the only cure for haemophilia2 but it is very costly. Haemophilia can be managed effectively with factor replacement therapy. This treatment involves injecting the missing clotting factor—Factor VIII for Haemophilia A or Factor IX for Haemophilia B—directly into the bloodstream. Treatment approaches include:
Prophylactic (Preventive) Therapy:
Regular injections of the clotting factor are administered to prevent bleeds, especially in children with severe haemophilia, who are at risk of spontaneous bleeding.
On-demand Therapy:
Injections are given in response to bleeding episodes such as joint bleeds that present with joint swelling, warmth or pain.
The dosage of Factor VIII or IX concentrate is determined by the child's weight and the severity of the bleed, and must be prescribed by a doctor/haematology specialist. Parents usually keep a supply at home for immediate use, with instructions on proper storage and administration provided for home therapy.
Children with hard-to-access veins may require a Portacath—a device implanted under the skin that allows easy access to a central vein for medication administration. The Portacath may need to be replaced if it becomes blocked or infected, and it typically lasts 2 to 3 years.
In cases of severe internal bleeding, such as brain or abdominal bleeding, emergency hospital treatment with high-dose factor replacement therapy is essential.
Inhibitors
Some children develop inhibitors—antibodies that neutralise the clotting factor, rendering replacement therapy ineffective. This occurs in approximately 20–30% of individuals with Haemophilia A3 and 1.5-3% with Haemophilia B4 undergoing factor replacement therapy. The risk of inhibitor development is linked to the type of genetic mutation present.
Managing haemophilia with inhibitors is challenging. For individuals with low inhibitors levels, higher doses of factor concentrate may be effective in controlling bleeding. However, patients with high levels of inhibitors require bypassing agents like recombinant activated Factor VII (Novoseven) or Factor Eight Inhibitor Bypassing Activity (FEIBA) for treatment of bleeds. Immune Tolerance Induction (ITI) therapy, involving daily or regular high-dose factor concentrate administration until inhibitors are eradicated, can also be used. ITI is successful in about 70% of patients with Haemophilia A5 ,typically taking 6 to 12 months to achieve success.
Emicizumab (Hemlibra), a bispecific monoclonal antibody given as a subcutaneous injection (injection into the fatty layer under the skin) is also used to prevent bleeds in haemophilia with inhibitors6.
Source:
2. Glasner MF, Pipe S, Miesbach W. Gene-based therapies for hemophilia. Res Pract Thromb Haemost. 2025;9(4):102870. Published 2025 Apr 25. doi:10.1016/j.rpth.2025.102870
3. Leissinger CA. Prevention of bleeds in hemophilia patients with inhibitors: emerging data and clinical direction. Am J Hematol. 2004;77(2):187-193. doi:10.1002/ajh.20162
4. DiMichele D. Inhibitor development in haemophilia B: an orphan disease in need of attention. Br J Haematol. 2007;138(3):305-315.
5. Antun A, Monahan PE, Manco-Johnson MJ, et al. Inhibitor recurrence after immune tolerance induction: a multicenter retrospective cohort study. J Thromb Haemost. 2015;13(11):1980-1988. doi:10.1111/jth.13143
6.Oldenburg J, Mahlangu JN, Kim B, et al. Emicizumab Prophylaxis in Hemophilia A with Inhibitors. N Engl J Med. 2017;377(9):809-818. doi:10.1056/NEJMoa1703068